Hydrogen bonds, best known for holding water and biomolecules together, now play a powerful role in solar energy conversion as part of a new supramolecular photocatalyst for water oxidation. Researchers from Inner Mongolia University and Tsinghua University report that carefully designed hydrogen bond interactions can reshape charge behavior in organic photocatalysts, opening a route to more efficient artificial photosynthesis.
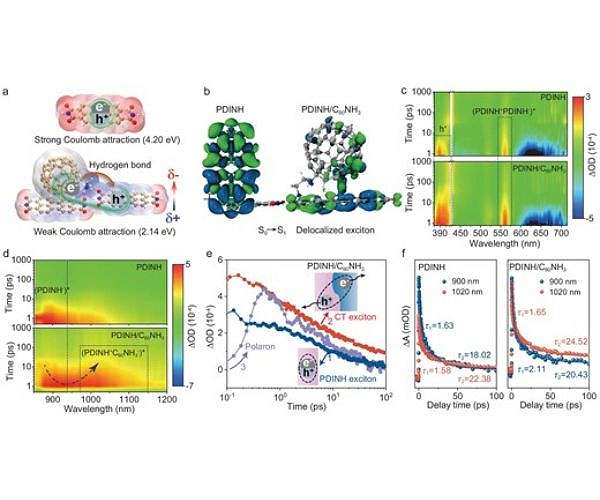
The team constructed a photocatalyst in which hydrogen bonds connect an electron donor, a perylene diimide supramolecule, to an electron acceptor, an aminated fullerene unit. These hydrogen bonds create a highly polarized local environment that improves dielectric shielding and weakens the Coulomb attraction between photogenerated electrons and holes. At the same time, the directional nature of the hydrogen bonds provides well-defined pathways that support exciton delocalization across the donor-acceptor interface.
By transforming tightly bound Frenkel-type excitons into weakly bound excitons for charge transfer, the hydrogen-bonded structure lowers the exciton binding energy and allows spontaneous dissociation of excitons under visible light. This spontaneous separation means that more of the absorbed photon energy appears as mobile charges that can drive redox chemistry rather than recombining as heat or light. The result is more effective use of photo-generated charges in the subsequent water oxidation reaction.
Compared to conventional supramolecular assemblies formed from single-component molecular building blocks, the hydrogen-bond designed donor-acceptor composite develops a much stronger internal electric field. This internal field arises from the strong electronic interactions at the interface and the asymmetric charge distribution imposed by the hydrogen bonds. The enhanced field sends electrons and holes in opposite directions, causing faster and more focused charge migration through the photocatalyst particles.
Under operating conditions, the researchers noted that the hydrogen-bonded system significantly increases the population of useful surface holes, which are the active oxidants in the splitting of water. After charge extraction and recombination processes were taken into account, the effective concentration of holes in the surface was increased by a factor of six compared to a comparable system without hydrogen-bonded interfaces. As more oxidizing holes reach the catalyst surface, the rate of the water oxidation half-reaction increases sharply.
In performance tests under visible light irradiation, the hydrogen-bonded photocatalyst achieved an oxygen evolution rate of 63.9 millimoles per gram per hour. The material also yielded an apparent quantum efficiency of 11.83 percent at 420 nanometers and 4.08 percent at 650 nanometers, indicating that it can use not only higher-energy blue light but also lower-energy red light to drive oxygen evolution. These figures place the system among the best reported organic photocatalysts for oxygen evolution under comparable conditions.
Most previous work on hydrogen bond-based photocatalysts has focused on promoting hydrogen evolution, hydrogen peroxide formation, or carbon dioxide reduction, with electron-driven reduction processes dominating. In contrast, the oxygen evolution reaction is the slower, kinetically demanding half-step of total water splitting, and progress in this area has been relatively slow. The new study shows that hydrogen bond engineering can be directly applied to this challenging oxidative step.
By demonstrating a hole-dominated organic semiconductor platform with state-of-the-art oxygen evolution performance, the work provides a design blueprint for constructing efficient general-purpose water splitting systems. It suggests that tuning the local electrostatic potential, exciton landscape, and internal electric field through supramolecular hydrogen bonds can provide a versatile handle for tuning charge dynamics. Such strategies can be extended to other organic or hybrid photocatalysts aimed at solar fuel production and related photoelectrochemical transformations.
Research report: Hydrogen bonding promoted the dissociation of excitons for efficient photocatalytic water oxidation