A Japanese-Chinese team developed a titanium molten salt redox flow battery, using abundant titanium ions and molten salt electrolytes to enable high-voltage, fast, and stable grid-scale energy storage. The system exhibits high efficiency of over 97%, strong cycle stability and improved cost and scalability compared to vanadium-based batteries, with further optimization underway.
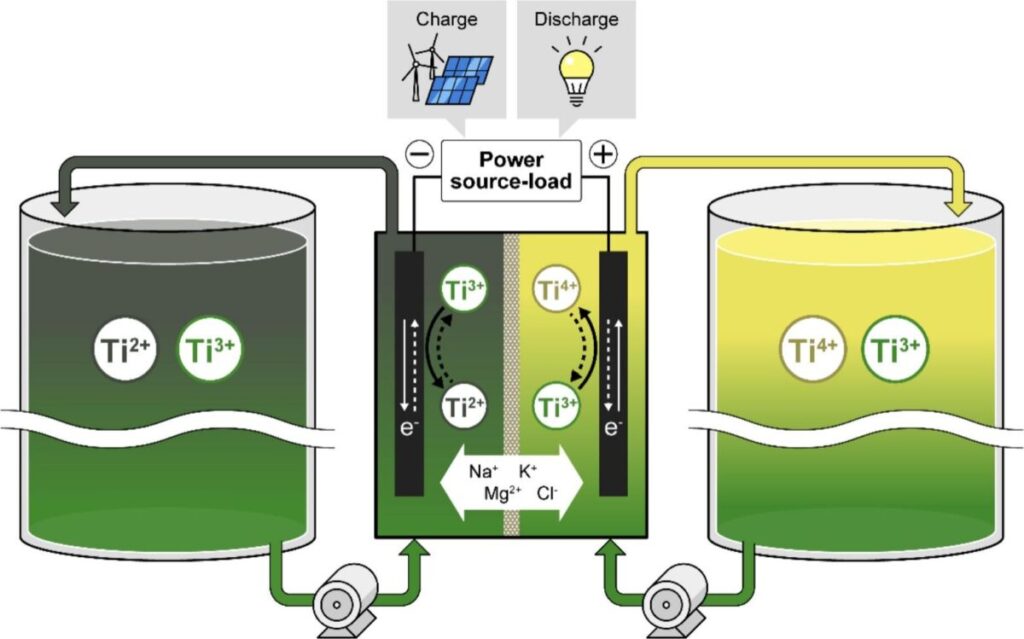
A Japanese-Chinese research team has titanium molten salt redox flow battery (TMSRB) that uses titanium ions as the redox active material and molten salt as the electrolyte.
The TMSRB is intended for use in grid-scale energy storage and is designed to provide higher charge-discharge current density compared to conventional vanadium redox flow battery (VRFB) designs.
The scientists explained that titanium, compared to vanadium, is a much more abundant element, addressing supply and cost constraints. “Titanium is the seventh most abundant metal in the Earth’s crust, with a crustal element abundance of 0.56%, 35 times that of vanadium. Thus, there is no concern about the sustainable supply of redox-active materials in TMSRB,” they pointed out.
The system works with titanium ions in multiple oxidation states, with the Ti⁴⁺/Ti³⁺ redox couple at the cathode and the Ti³⁺/Ti²⁺ redox couple at the anode, allowing reversible redox reactions. It also uses molten salt electrolytes, such as lithium chloride-potassium chloride (LiCl-KCl) and sodium chloride-magnesium chloride-potassium chloride (NaCl-MgCl₂-KCl), which the scientists said provide a wide electrochemical stability window and high ionic conductivity, while supporting efficient operation and high-voltage operation, fast charge-discharge rates and stable cycling at temperatures between 300 and 450 C.
The battery also uses a porous aluminum oxide (Al₂O₃) crucible as a separator, along with carbon and graphite electrodes connected by nickel cables. Titanium tetrachloride (TiCl₄) is carefully introduced into the system and its evaporation is controlled using lithium fluoride (LiF) additives.
The battery was assembled with the separator placed between the positive and negative compartments. The negative electrode consisted of a multilayer carbon net connected to a graphite rod as a current collector and a nickel rod as a lead. On the other side, a similar configuration was used, including a hollow carbon cylinder. The molten salt electrolytes containing titanium ions were added to the cell and LiF was introduced on the positive side to suppress the evaporation of TiCl4.
The fully assembled battery was then operated in a resistance furnace under an argon (Ar) atmosphere to evaluate its redox behavior and charge-discharge cycle stability. In addition, ab initio molecular dynamics (AIMD) simulations were used to monitor the ion distribution during operation.
The analysis demonstrated the suitability of the multivalent titanium ions as redox-active materials for high-performance batteries. Cyclic and square wave voltammetry in molten LiCl-KCl at 400 C revealed bright and reversible Ti²⁺/Ti³⁺ and Ti³⁺/Ti⁴⁺ redox reactions, which the scientists said could yield a high theoretical cell voltage of around 1.55 V, extendable to 1.80 V if Ti/Ti²⁺ is included. Moreover, multiple stable oxidation states and various redox transitions were found to further improve system flexibility and stability.
The researchers also found that the composition of the molten salt can be tuned to optimize cost, temperature range and electrochemical performance. Experiments with different electrolytes confirmed consistent redox activity and high voltage over a wide temperature range.
Furthermore, experimental tests have demonstrated high coulombic efficiency of over 97% and a “stable” cycle even at high charge-discharge rates. The performance of several molten salt systems remains strong, highlighting their robustness and adaptability.
“In summary, the developed TMSRB has great advantages, especially higher operating voltages, extremely high coulombic efficiency, fast charge-discharge capabilities and abundant, cheap raw materials,” the academics pointed out. “Further technical optimization, such as advanced cell stack designs, improved thermal management strategies and more detailed evaluations of system-level performance metrics, including voltage efficiency, energy efficiency, electrolyte tank capacity and practical volumetric energy density, is currently underway.”
The VRFB system was introduced in “A powerful redox flow battery for grid-scale energy storage”, published in Electrochemical communication. The research team included academics from the University of Science and Technology Beijing in China and Tohoku University in Japan.
This content is copyrighted and may not be reused. If you would like to collaborate with us and reuse some of our content, please contact: editors@pv-magazine.com.