Scientists in China have built and tested a device that uses redox couples combined with a single three-junction amorphous-silicon photoelectrode. When tested under a xenon lamp simulating a single sun, the device achieved an average solar energy to electricity conversion efficiency of 4.2%.
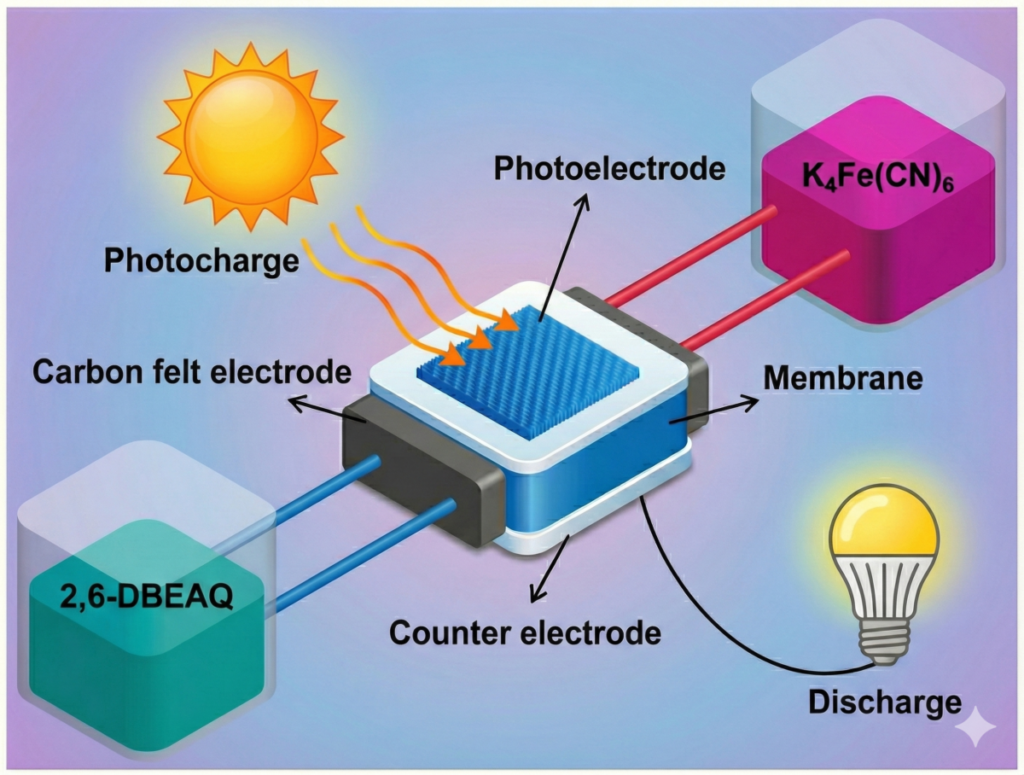
A research team from China’s Nanjing Tech University (NanjingTech) has developed and built a new anthraquinone-based solar redox flow battery (SRFB) device.
The system consists of redox couples known as 2,6-DBEAQ and K4[Fe(CN)6]combined with a single three-junction amorphous silicon photoelectrode. SRFBs are systems that combine a solar cell with a redox flow battery, allowing simultaneous sunlight capture and chemical energy storage.
“Among the redox couples used in SRFBs, aqueous organic anthraquinone derivatives have gained recognition as the favored materials for energy storage,” corresponding author Chengyu He shared. pv magazine. “Anthraquinone-based SRFBs typically employ redox couples such as AQDS and 2,6-DHAQ, which operate in both strongly acidic and alkaline conditions. However, due to the corrosion of photoelectrodes and the instability of the paired redox couple, these SRFB devices exhibit relatively low solar electricity efficiency.”
For the photocathode portion of the device, the group cut commercial amorphous silicon (3jn-a-Si) photovoltaic cells with three junctions into 2 cm x 2 cm pieces. The cell consists of stacked compounds of amorphous silicon-germanium alloys deposited on a stainless steel substrate covered with a layer of indium tin oxide (ITO). It is electrically connected to a carbon felt counter electrode via an external circuit.
The photocathode is in chemical contact with the catholyte containing 2,6-DBEAQ, which is reduced during photocharging. In contrast, the anolyte containing K₄[Fe(CN)₆] is oxidized at the carbon felt electrode. The two electrolytes flow continuously from external tanks through the cell using a peristaltic pump. They are kept separated by a Nafion ion exchange membrane that allows ion transport to maintain charge balance but prevents mixing of electrolytes.
The performance of the SRFB device was evaluated by a cyclic test, a repeated charge-discharge experiment. Before measurements, the electrolytes were flushed with argon for 30 minutes to remove dissolved oxygen. During charging, the photocathode was irradiated with a xenon lamp simulating the intensity of 1 sun at 100 mW cm2. The device was discharged at a current density of 10 mA cm-2 until the battery voltage dropped to 0.2 V.
“The SRFB device can be charged and discharged in ten cycles without external bias, while the electricity efficiency of solar energy is up to 4.3%,” said Chengyu He. “The successful preparation of this SRFB device opens new opportunities for the further development of advanced solar-to-chemical energy conversion technologies.”
The results are presented in “An aqueous anthraquinone solar redox flow battery for efficient solar energy conversion and storage”, published in Electrochimics Acta.
This content is copyrighted and may not be reused. If you would like to collaborate with us and reuse some of our content, please contact: editors@pv-magazine.com.