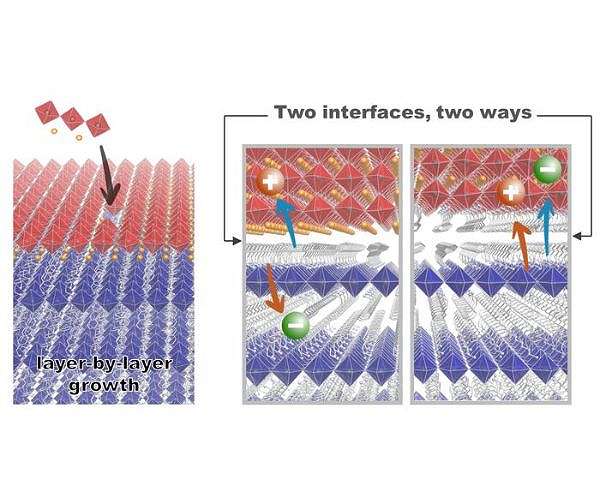
Researchers have achieved a new level of control over the atomic structure of a family of materials known as halide perovskites, creating a finely tuned ‘energy sandwich’ that could transform the way solar cells, LEDs and lasers are made.
Because of their remarkable ability to absorb and emit light, and because they are cheaper and can be configured to convert a greater portion of the solar spectrum into energy than silicon, perovskites have long been touted as a potential replacement for silicon in solar cells, LEDs and quantum technologies.
However, their instability and durability have largely limited perovskite devices to the laboratory until now. In addition, scientists have struggled to precisely control the thickness of perovskite films, and to determine how different perovskite layers interact when stacked on top of each other – an important step in building functional, multilayer structures.
Now, a team of researchers led by the University of Cambridge has found a new way to grow ultra-thin layers of perovskite films so that their atoms align perfectly, which could enable more powerful, durable and efficient devices.
The researchers used a vapor-based technique to grow three-dimensional and two-dimensional perovskites layer by layer, allowing them to control the thickness of the films down to fractions of an atom. Their results, reported in the journal Science, could open the door to useful perovskite devices that can be produced on a large scale, using a process like that used to make commercial semiconductors.
Each layer in a semiconductor “sandwich” does a different job in moving electrons and their positively charged counterparts (called holes) and determines how the semiconductors absorb or emit light. Together, the layers act as one-way streets that conduct the electrical charges in opposite directions, preventing them from colliding with each other and wasting energy in the form of heat.
In other commonly used semiconductors, such as silicon or newer materials such as gallium nitride, the properties of the individual layers can be refined in various ways. But perovskites, despite their excellent performance, have so far proven difficult to control in layered devices, partly because of their ‘chaotic’ atomic structure.
“A lot of perovskite research uses solution processing, which is messy and difficult to control,” says Professor Sam Stranks from the Department of Chemical Engineering and Biotechnology, who co-led the research. “By switching to vapor processing – the same method used for standard semiconductors – we can achieve the same degree of atomic control, but with materials that are much more forgiving.”
The researchers used a combination of three-dimensional and two-dimensional perovskites to create and control their atomically tuned stacks, a phenomenon known as epitaxial growth. This fine control allowed the team to directly observe how the light emitted by the material changes depending on whether it is a single layer, a double layer or thicker.
“The hope was that we could grow a perfect perovskite crystal where we could change the chemical composition layer by layer, and that’s what we did,” says co-first author Dr Yang Lu from Cambridge’s Department of Chemical Engineering and Biotechnology and Cavendish Laboratory. “It’s like building a semiconductor from scratch, one atomic layer after another, but with materials that are much easier and cheaper to process.”
The researchers also found that they could engineer the connections between the layers to determine whether electrons and holes stayed together or apart — a key factor in how efficiently a material emits light.
“We’ve achieved a level of tunability that wasn’t even on our radar when we started,” says Professor Sir Richard Friend of the Cavendish Laboratory, who co-led the research. “We can now decide what kind of node we want – one that holds charges together or one that pulls them apart – just by slightly changing the growing conditions.”
The researchers found that they could reduce the energy difference between the layers by more than half an electron volt, and in some cases extend the lifetime of electrons and holes to more than 10 microseconds: much longer than normal.
The team says this level of precision could pave the way for scalable, powerful devices that use light in new ways, from lasers and detectors to next-generation quantum technologies.
“Changing the composition and performance of perovskites at will – and investigating these changes – is a real achievement and reflects the amount of time and investment we have made here in Cambridge,” said Stranks. “But more importantly, it shows how we can make working semiconductors from perovskites, which could one day revolutionize the way we make low-cost electronics and solar cells.”
The research was supported in part by the Royal Society, the European Research Council, the Simons Foundation and the Engineering and Physical Sciences Research Council (EPSRC), part of UK Research and Innovation (UKRI). Richard Friend is a Fellow of St John’s College, Cambridge. Sam Stranks is a Fellow of Clare College, Cambridge.
Research report:Layer-by-layer epitaxial growth of perovskite heterostructures with tunable band offsets